583g Catalytic Deoxygenation of Stearic and Oleic Acids Over Pd/C
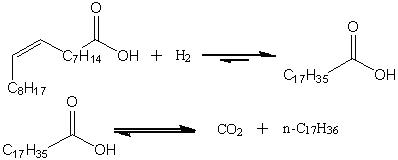
Catalytic deoxygenation of C18 free fatty acids (FFAs) can be used to produce C17 hydrocarbons as an alternative to conventional fatty acid methyl ester (FAME) biodiesel [1]. FFA deoxygenation occurs via two pathways: decarbonylation and decarboxylation. In this work, deoxygenation of oleic and stearic acids over a 5 wt.% Pd/C catalyst was investigated to determine the effects of unsaturation of the alkyl chain and the feasibility of using the hydrocarbon reaction product as solvent. FFA deoxygenation was conducted in a semi-batch autoclave at 300ºC and 15 atm using either n-dodecane or n-heptadecane as solvent with a constant flow of 60 mL/min of He or 10% H2 in He. Deoxygenation of stearic acid occurs primarily via a decarboxylation pathway in either He or 10% H2 flow. The initial decarboxylation rate under He is much higher than under 10% H2, but the reaction reaches completion more quickly in the latter case because H2 inhibits catalyst deactivation. Deoxygenation of oleic acid is strongly inhibited under He, achieving a conversion of only 12% after 3 h with less than 10% selectivity to heptadecane. In contrast, the oleic acid deoxygenation goes to completion in <1 h in the presence of H2 yielding n-heptadecane in essentially 100% yield. Additional experiments established that the overall reaction for oleic acid is a stepwise process: initial hydrogenation of the double bond to form stearic acid and subsequent decarboxylation to n-heptadecane. Using n-heptadecane as solvent, the initial rate of stearic acid decarbonylation under 10% H2 was equivalent to that observed in n dodecane. The initial rate of decarboxylation, however, was substantially lower due to reversibility making decarbonylation a more significant pathway in the overall reaction. Stearic acid deoxygenation nears completion in n-heptadecane in ~2 h aided by removal of the CO2 reaction product.